Upcoming products & services
Our strategic development pipeline includes the automation and miniaturisation of multiplex assays to read amplification in colorectal cancer - derived exosomes in blood; early detection of colorectal cancer using microbiota and methylation tests; and innovate programs for Lifestyle interventions to prevent disease
The merge and optimisation of microfluidic systems, automated bead-droplet workflow and multiplex bead assays, to capture and characterise exosomes is aimed at detecting colorectal tumours from a blood samples. The proposed novel approach allows isolation of exosomes from a clinically feasible volume of blood, amplifying sensitivity while maintaining specificity (M3 Profiler).
Products
current and pipeline
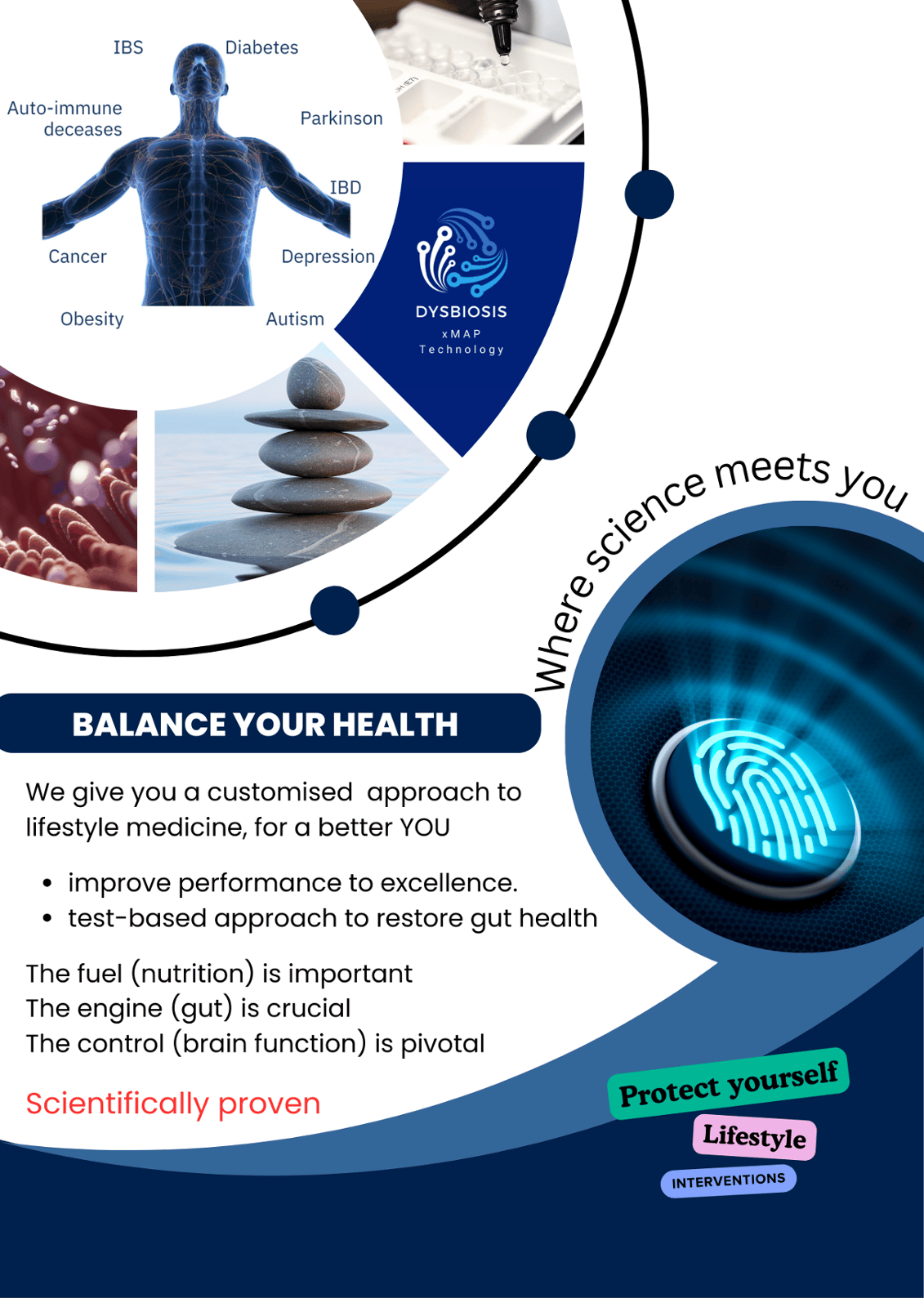
CE-IVD Dysbiosis test
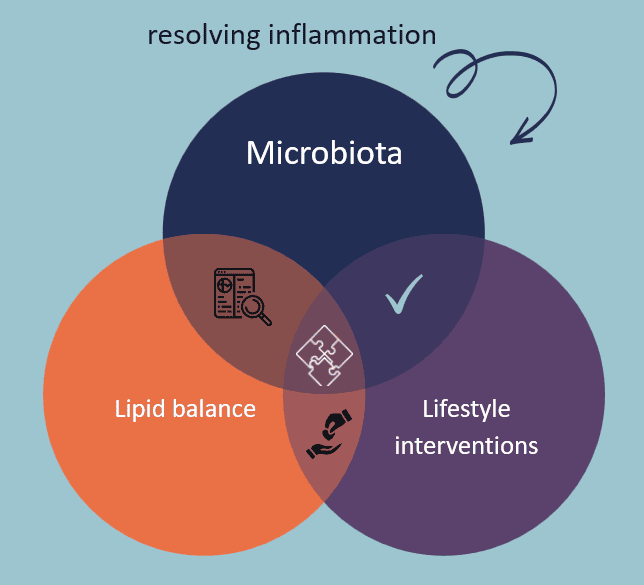
Dysbiosis test using a fecal sampler. Quantification of 50 bacterial families to generate a dysbiosis index and functional profiles assisting personalised interventions. The tests provides a diagnosis of inflammatory bowel disease and identifies the cause of intestinal inflammation related to metabolic diseases, irritable bowel syndrome and other conditions.

Colorectal Cancer (CRC) Detect
The CRC Detect is a real-time PCR test measuring the methylation state of genes that are associated with early detection of colorectal cancer. DNA is extracted from a fecal sample and the analysis is done in a licensed clinical laboratory.
The result indicates the presence of early disease (early adenomas) and supports colonoscopy results. The test should be recommended to FIT positive individuals that are not compliant with the colonoscopy procedure. A positive result shall be confirmed.
Omnigene Medical Technology Ltd partnered with academic and biotech companies to expand the line of research, including early detection of colorectal cancer, measuring pre-lesions of cervical and colorectal carcinoma, dysbiosis (imbalance in microbiota) testing towards measuring intestinal inflammation and many more.
Our Pricing Plan
We partner with our customer to provide the best products and services.
Contract Research
Pricing per project basis
CE-IVD
Dysbiosis test
Euro 295
Dysbiosis test
Euro 295
Breast Cancer classification test
Ask for
sales distributor